The best way is to periodically (eg every few months or so) do a capacity test by running the UPS (from fully charged, with power off) with a known heavy load and recording the time it takes to drop out on low Volts.
The important thing is that you can graph the results over time (eg years) and note how it is deteriorating. This will give you notice well in advance of any failure due to low capacity.
If you can't do a full capacity test, simply measure the instantaneous voltage drop with a known large load, and again graph the results.
Terminal voltage by itself is not at all useful.
Your point can be very easily made differently.
If you look at the discharge curve for a Lead-Acid Battery with a 12V or 6V rating:
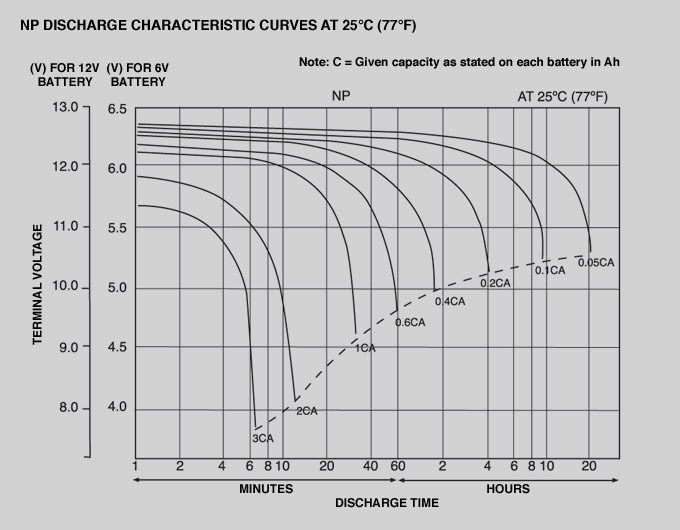
This comes from Yuasa. They make the things. It's either reliable or optimistic, certainly not pessimistic.
Let's look at the 12V one and optimistically assume that you are only interested in 0.2C discharge, any other rate the same arguments can be made with a different line.
At its 12V mark, you can see there is a "tipping point" where the voltage goes from relatively constant to plummeting.
At 11V it is almost going straight down.
At 9V it will drop right away under the same loading current.
This means that at the 12V point you have essentially used 60% of its capacity. At 11V you are at effectively 93%. At 9V you're at 99.5%.
Now adding that a battery with 0V across it has no chemical initiative any more, of any chemistry type, you will need to motivate it re-create the chemical imbalance that causes the apparent voltage and potential for current to flow. This is hard in any battery (which is why almost all batteries are built with electrodes already chemically built of the right materials to create the cell potential).
With SLA a lot of energy goes in chemical recombination of Sulphates and Sulphites, wasting a lot of energy. This results in needing excessive power to re-engage a cell that is left at 0V. Excessive power = heat. Heat = gassing. Gassing = moisture loss. Moisture loss = bad. Not to mention the higher voltage usually required makes many, many by-products on the plates, next to by-products already generated by neutering it in the first place and you're left with a AA battery with the weight and size of a 6Ah SLA.
Now, if you go near 9V, it will become 0V very quickly. You'll easily be too late.
Many manufacturers tell us "Consider your battery empty at 11.8V", some people (me included) assume 11V. Those who use 11V often (but not always) take care to know that this is the lower limit. This is exactly for that reason.
If they will not accept that 100's of manufacturers, countless experts in the field and users alike say "at 11.8V you're not going to get much more out of it and it'll be risky to try", then just convince them with the fact that at 11V there won't be much to get any more anyway. Done. All other points moot.
The reason a car battery can be dropped to 2V and then keep working, is because that battery was at 2V very shortly, because the idiot leaving his lights on realised after a while. And because they are usually over dimensioned by a factor of 2 to 5, depending on the type and brand of a car, so a crippled one will work for a couple more years.
And let's be honest, are we really going to put hours and hours of work and research into convincing someone who can't even manage to mind his car lights of agreed fact shared between thousands of electrical engineers worldwide?

Best Answer
The Battery Council International (BCI) created a group of standardized specifications for lead-acid batteries including Reserve Capacity. This particular test gave consumers a useful comparison for a lead-acid battery. Measuring the full discharge in minutes is the reserve time a consumer could expect with a new battery after an alternator failure and a standard 25A constant drain.
Although I was not aware of this standard test until this question, I agree it is quicker, reliable and more accurate than leaving all your vehicle lights on and hoping it will start in the morning.
Although it may not be the criteria used for warranty purposes or factory acceptance testing, I agree it is an important consumer test for State of Health (SOH). The ratio to the original battery spec or similar replacements is a valid metric.
Reason:
Reserve Capacity, RC is not a test you want to do often, nor is it practical for manufacturers to spend this much time but it is relatively easy to perform.
Here are two different priced batteries comparing their specs from one supplier for Absorbed Glass Mat (AGM) 12V batteries.
RC: RESERVE CAPACITY in minutes for a fully-charged battery to supply 25 amps at 80°F (28'C), until the battery is discharged down to 10.5 volts (1.75V/cell)
RC: how long a fully-charged battery can deliver 25 amps of current in an 80°F environment before the battery is discharged down to 10.5 volts (1.75V/cell)
Reserve capacity, RC is the number of minutes a battery can maintain a useful voltage with a constant 25A drain from a full charge.
AMP-HOUR Rating. This is battery capacity and is measured by the 20-hour constant current product from 100% to 0% in 20 Hrs.
CCA is the maximum amperes sustained for 30 sec. when 12.5V battery with Sp. Gr.= 1.255 - 1.260 is loaded down to 7.5V at 0°F. This applies to starter batteries.
(Thanks for the question, I learnt something new)